
Biopolymers have special properties that distinguish them from other polymers. These three biopolymers, the subject of this paper, are polynucleotide (DNA and RNA), polypeptide (protein), and polysaccharide (polymerized sugars). Living systems are united by their expression and utilization of three types of polymers. Polyethylene, for example, is a synthetic polymer with molecular formula (–C 2H 4–) n and molecular weight around 5 million Daltons that is used to make plastic bottles and bags. Polymers are large molecules formed by covalently linking small monomers into chains. We believe that an understanding of the structure, function, and origins of any given type of biopolymer requires the context of other biopolymers. These universal properties support a model in which life’s polymers emerged simultaneously and co-evolved in a common hydrolytic milieu where molecular persistence depended on folding and assembly. Heterogeneous association achieves partner-preservation. 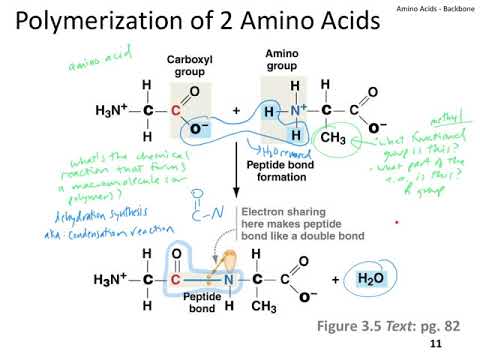
Self-association in a hydrolytic environment achieves self-preservation. These protected states are folded, using self-complementary interactions among recurrent folding elements within a given biopolymer, or assembled, in associations between the same or different biopolymer types. All three biopolymers can access specific states that protect against hydrolysis. All three biopolymers are synthesized by dehydration reactions that are catalyzed by molecular motors driven by hydrolysis of phosphorylated nucleosides. We observe that all three biopolymer types are pre-organized, conditionally self-complementary, chemically unstable in aqueous media yet persistent because of kinetic trapping, with chiral monomers and directional chains. We incorporate this information into a model that explains much about their origins, selection, and early evolution. Here we evaluate both universal and idiosyncratic characteristics of these biopolymers. The modification occurs after the amino acid has been assembled into a protein.Life as we know it requires three basic types of polymers: polypeptide, polynucleotide, and polysaccharide. In some cases an amino acid found in a protein is actually a derivative of one of the common 20 amino acids (one such derivative is hydroxyproline). Glycine, the major amino acid found in gelatin, was named for its sweet taste (Greek glykys, meaning “sweet”). It was obtained from protein found in asparagus juice (hence the name). The first amino acid to be isolated was asparagine in 1806. The only amino acid whose R group has a pK a (6.0) near physiological pHĪlmost as strong a base as sodium hydroxide Oxidation of two cysteine molecules yields cystineĪmino acids with a negatively charged R groupĬarboxyl groups are ionized at physiological pH also known as aspartateĬarboxyl groups are ionized at physiological pH also known as glutamateĪmino acids with a positively charged R group Named for its similarity to the sugar threose Side chain functions as a methyl group donorĬontains a secondary amine group referred to as an α-imino acidĪmino acids with a polar but neutral R group The only amino acid lacking a chiral carbonĪn essential amino acid because most animals cannot synthesize branched-chain amino acidsĪlso classified as an aromatic amino acid \): Common Amino Acids Found in Proteins Common Name
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
